PyramIDH Clinical Trial
About the PyramIDH Trial
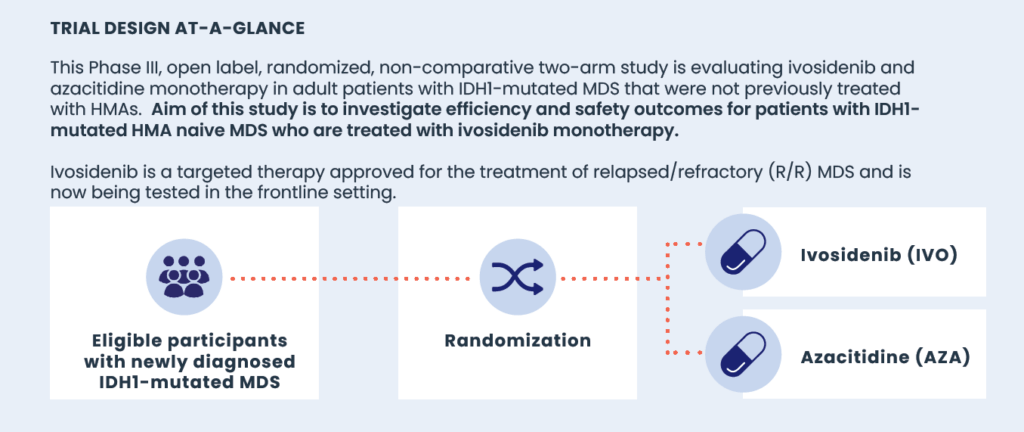
What is the PyramIDH Trial?

Patients with isocitrate dehydrogenase 1 (IDH1)-mutated newly diagnosed MDS have increased risk of transformation to AML, worse event-free survival (EFS), and reduced OS when compared with patients with wild-type IDH1. Treatment options for such patients are limited. The Phase III PyramIDH clinical trial, sponsored by Servier, is investigating ivosidenib, an oral, targeted inhibitor of mutant IDH1 enzymes, to assess its efficacy and safety as a potential new treatment for this patient population.
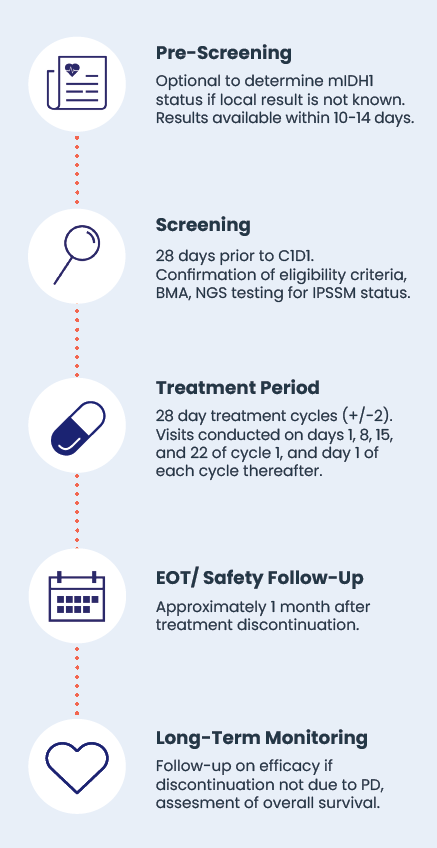
Important Attributes
Study Objectives
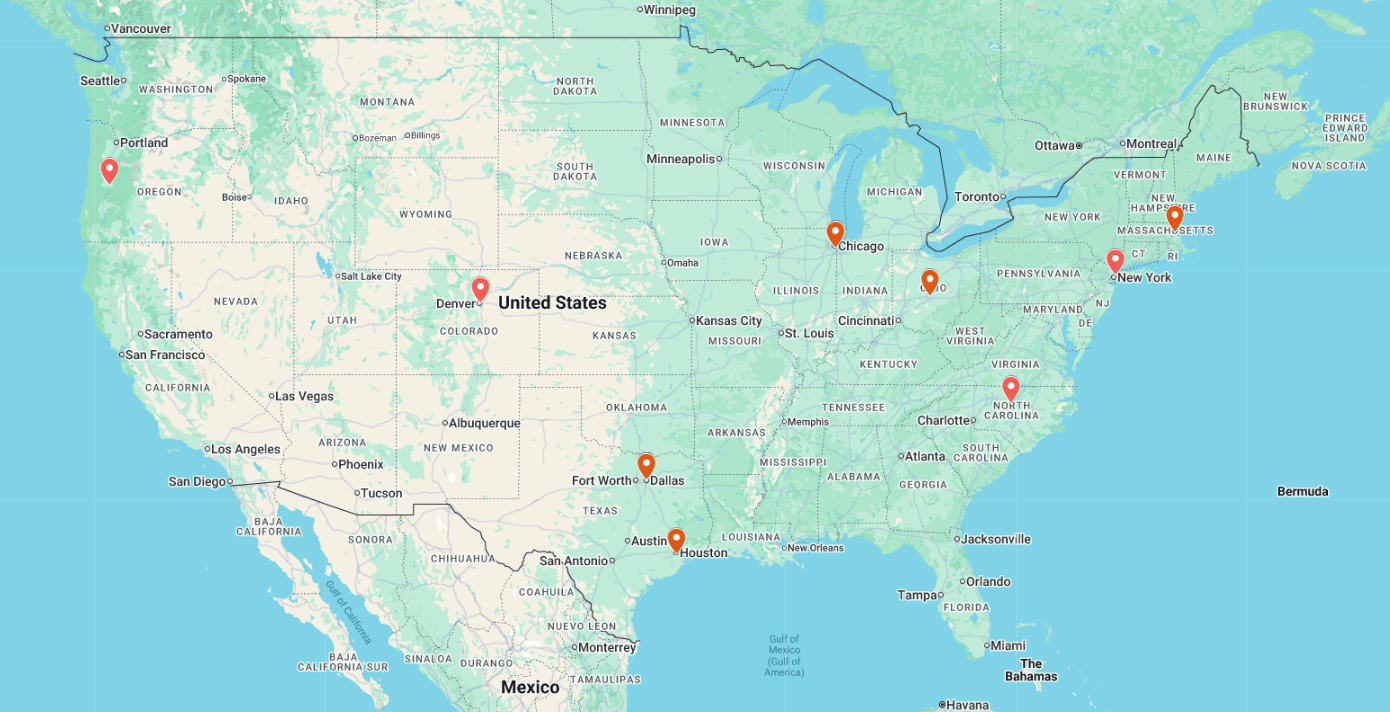
| Site Locations: | Principal Investigator |
| Massachusetts General Hospital | Dr. A. Brunner |
| OSU Comprehensive Cancer Center | Dr. U. Borate |
| MD Anderson Cancer Center | Dr. G. Garcia-Manero |
| UT Southwestern Medical Center | Dr. Y. Madanat |
| University of Chicago Medicine | Dr. T. Odenike |
| Memorial Sloan Kettering Cancer Center New York – Main Hospital | Dr. T. Haque |
| UNC Lineberger Comprehensive Cancer Center | Dr. Williams |
| Willamette Valley Cancer Institute and Research Center – Eugene | Dr. L. Fletcher |
| Colorado Blood Cancer Institute | Dr. M. Rotta |
To refer a patient to a participating site, please contact the nearest site to you or Servier at scientificinformation@servier.com.
US-MED-01076 03/05/26